 
In humans, several diseases are due to the accumulation of misfolded or unfolded proteins. Misfolding of proteins has detrimental effects on the overall functioning of the cell. Most proteins go through several intermediate forms before folding into the most stable, biologically active structure. Hence the specific activity of each protein depends on its unique three-dimensional architecture.įor a protein to be functional, it must fold accurately. A protein’s function mostly depends on its ability to recognize and bind other molecules, analogous to a lock and key. Proteins perform a wide range of biological functions such as catalyzing chemical reactions, providing immunological defense, storage, transport, cellular communication, movement, and structural support. 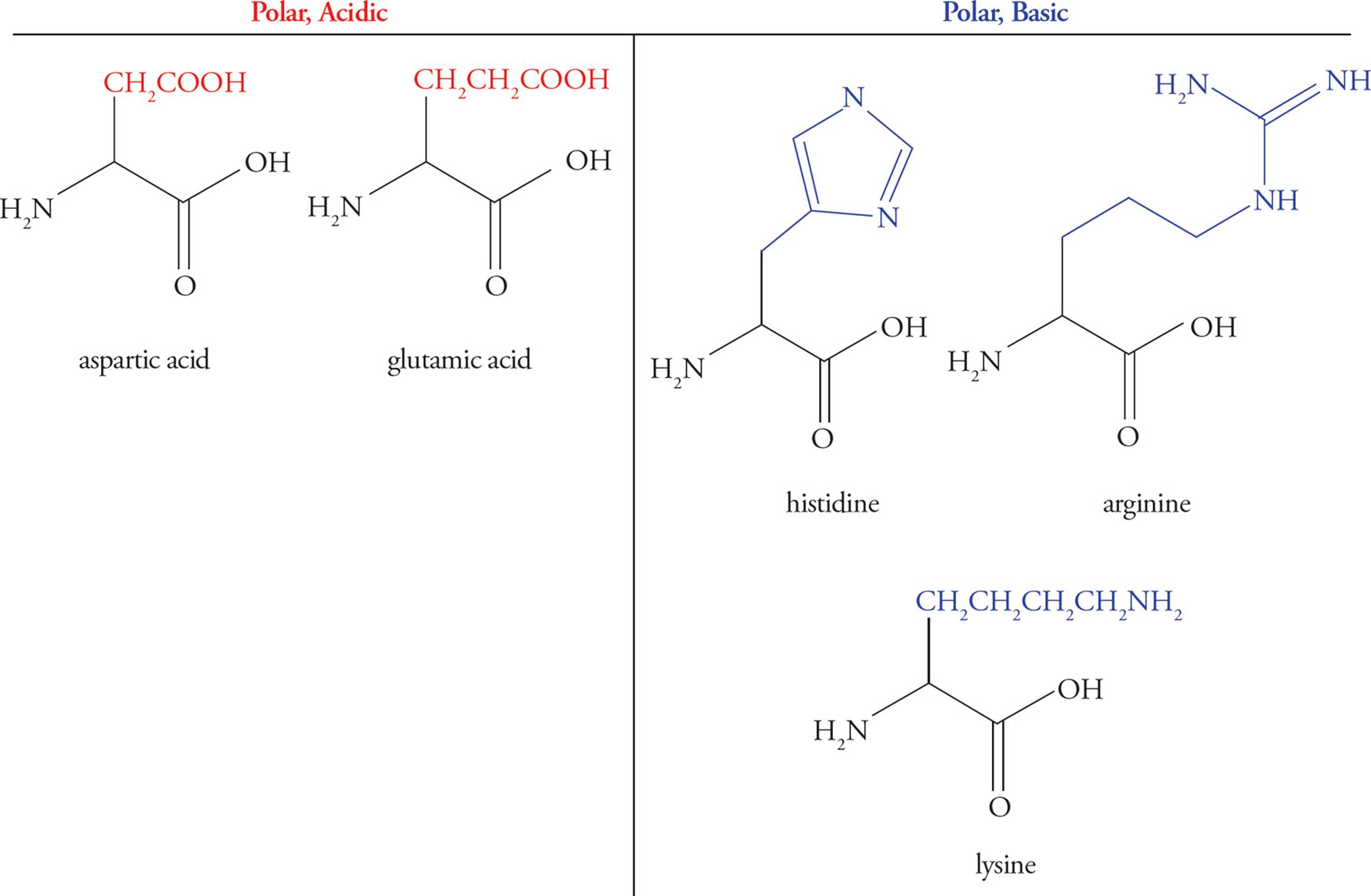
Protein Structure Is Critical to Its Biological Function 
Interactions between its constituent amino acids guide protein folding, and hence the protein structure is primarily dependent on its amino acid sequence. Upon synthesis, a protein folds into a three-dimensional conformation which is critical to its biological function. Proteins are chains of amino acids linked together by peptide bonds. The presence of these chemical interactions and bonds secure the protein in its most favored confirmation. These bonds occur between two adjacent cysteine monomers, which contain sulfhydryl or SH groups on their side chains. The sulfur on one bonds covalently to the second. Lastly, acting as reinforcement are disulfide bridges. Polar side chains can form hydrogen bonds, either with water or other polar molecules. Side chains with opposite charges can form ionic bonds, while similarly charged sides repel each other. On the outside mostly lie the amino acids with charged or polar side chains, free to interact with water. Weak van der Waals attractions help maintain the clustered core. First, the compactness is governed by hydrophobicity of the amino acid side groups, such that non-polar chains are pushed inside creating a hydrophobic core away from the aqueous environment. More specifically, the folding patterns are determined by different chemical interactions. After the formation of its secondary structure, a protein collapses into a tertiary globular form, a unique and individual 3D confirmation that guides its function. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
